PCO-T-01
LYCRA TLSO ORTHOSIS WITH SOFT BACK PANEL
Our TLSO orthosis PCO-T-01 increases stabilization and body awareness in case of neurological disorders.
HOW DOES IT WORK?
Made of soft PowerElastic™, provides deep, core compression and normalizes muscles tone.
PowerElastic™ is a very thin and very flexible fabric for the production of delicate and sophisticated orthopedic products. It consists of spandex, the action of which is responsible for proper compression and fitting of the product to the patient's body, and reduces muscle vibrations generated during increased physical exertion. It is responsible for maintaining the proper climate next to the skin and effective drainage of sweat, and its structure guarantees velvety softness to the touch. PowerElastic™ is skin-friendly fabric and has the Oeko-Tex Standard 100 certificate.
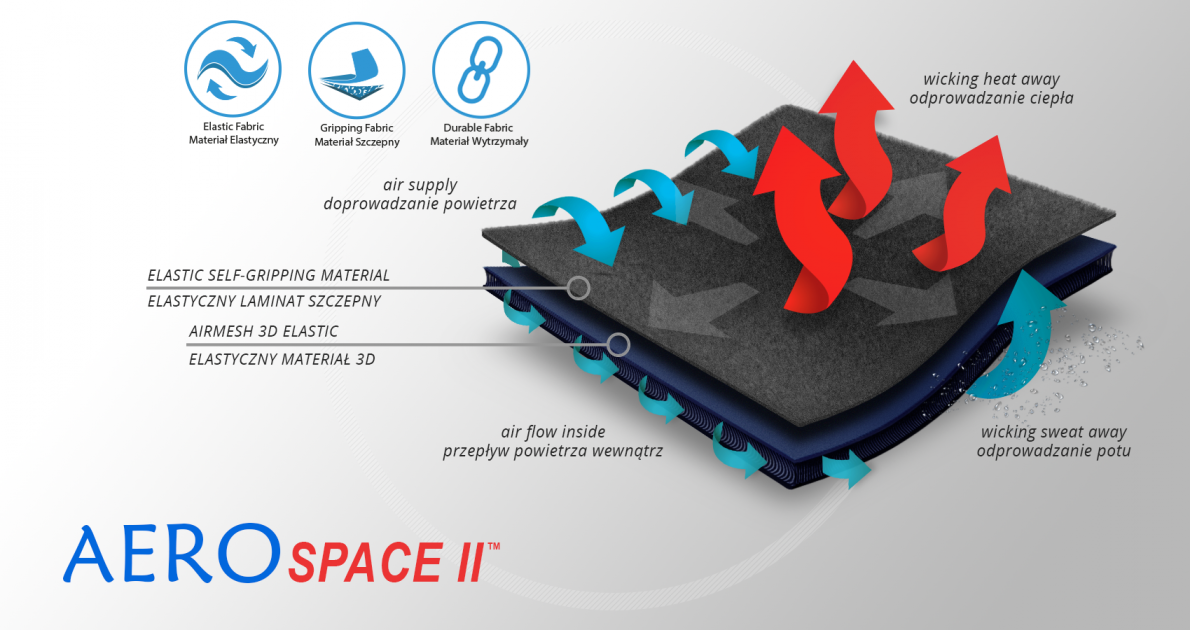
The back of the brace is made of AeroSpace II ™ material
AeroSpace II™ is an innovative new generation raw material. It is a distance elastic 3D knitted fabric consisting of two layers of facings and an interlacing that creates the appropriate thickness of the raw material and has relieving properties. The knitwear is made of the highest quality polyester yarn - guaranteeing high mechanical strength and spandex ensuring its proper flexibility. AeroSpace II™ has an external self-gripping layer what makes it easy to adjust each product to individual patient’s needs. This material is characterized by a very low weight, high flexibility and a very large openwork structure, allowing for very easy drainage of sweat from the body and bringing fresh air to the skin. Products made of this raw material are neutral to the secured joint, do not heat or cool it, but ensure its proper compression and fit and reduce muscle vibrations generated during physical exertion. Its thickness and 3D structure perfectly relieves the orthopedic splints, stays or other elements mounted on the product and guarantees velvety softness to the touch.

The back panel provides additional stabilization and reduces the pressure of bone prominences. Elasticity and deep pressure improves body coordination and limits the involuntary movements.
Our PCO-T-01 dynamic suite with back panel should be used as an addition to occupational and physical therapy activity. This solution will reduce involuntary movements, normalize muscle balance and improve therapeutic effects.
The back panel maintains the body temperature, reduces pain and improves healing. What is more, the compression provides proprioceptive input that is necessary in case of neurological disorders.
It is believed that this type of Postural Compression Orthoses PCO® and deep compression provides better sensory information to the brain, increases proprioception and child’s knowledge about its own body. Due to this fact, dynamic PCO® orthoses are the best solution for improving treatment in case of abnormal tone and decreasing spasticity.
BENEFITS
- improves body balance
- increases core stability
- increases passive and active ROM
- normalizes muscle tone
- decreases spasticity
- decreases involuntary movements
- improves breathing
INDICATIONS
- cerebral palsy (CP)
- spina bifida
- Down Syndrome
- sensory integration (SI)
- high tone
- spasticity
- low tone
- atetosis
- ataxia
- dyskinesia
- dystonia
Available sizes
| Size | Torso circumference (A) | Trochanter greater to under-arm length (B) | How to measure |
|---|---|---|---|
| 1 | 40 – 50 cm | 17 – 21 cm | 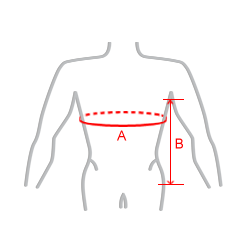 |
| 1 MAXI | 40 – 50 cm | 21 – 27 cm | |
| 2 | 48 – 65 cm | 21 – 27 cm | |
| 2 MAXI | 48 – 65 cm | 27 – 31 cm | |
| 3 | 60 – 70 cm | 27 – 31 cm | |
| 4 | 60 – 70 cm | 31 – 35 cm | |
| 4 MAXI | 60 – 70 cm | 35 – 40 cm | |
| 5 | 68 – 80 cm | 35 – 40 cm |
Total height of the product:
| Size | Back | Side |
|---|---|---|
| 1 | 27 cm | 19 cm |
| 1 MAXI | 31 cm | 22 cm |
| 2 | 31 cm | 22 cm |
| 2 MAXI | 34 cm | 26 cm |
| 3 | 36 cm | 26 cm |
| 4 | 39 cm | 30 cm |
| 4 MAXI | 44 cm | 34 cm |
| 5 | 44 cm | 34 cm |
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES..
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.







 Class I medical device in accordance with the Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices.
Class I medical device in accordance with the Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices.