LYMPHEDEMA AND POST-TRAUMATIC EFFUSIONS
Lymphedema is a chronic accumulation of lymphatic fluid within the tissues, resulting from impaired lymphatic drainage. In the foot and ankle region, it causes a feeling of heaviness, skin tightness, and an increase in limb circumference. Regular, well-adjusted compression is a key element of therapy, helping to control swelling, support lymphatic transport, and prevent the progression of edema.
In the case of post-traumatic effusions (e.g., following an ankle sprain or ligament injury), damaged blood and lymphatic vessels lead to fluid accumulation within the soft tissues. This results in swelling, pain, and reduced joint mobility. Compression limits the formation of effusion and subsequently supports its absorption, accelerating recovery and return to full function.
The AM-OSS-16 ankle support compression brace was created specifically to effectively support the lymphatic and circulatory systems within the lower limb.
Product characteristics:
The AM-OSS-16 foot and ankle compression brace is a modern, practical, and comfortable solution for individuals requiring targeted compression therapy in the foot and ankle area. Thanks to its adjustability, comfort of use, and ability to fit the limb individually, the product combines therapeutic effectiveness with everyday convenience. It improves lymphatic and venous flow, prevents fluid accumulation, and reduces swelling, enhancing mobility and overall comfort of life.
The AM-OSS-16 ankle brace is designed to provide therapeutic, adjustable compression to the foot and ankle. It is an excellent solution for people dealing with lymphedema, circulation disorders, or post-injury effusions of the ankle joint.
Thanks to its lightweight and elastic structure, the brace conforms perfectly to the shape of the foot, while adjustable straps allow the user to control the level of compression as needed. This precise fit makes it a comfortable alternative to traditional compression bandages, enabling quick and independent application.
The unique UniPren™ material, developed through years of research and experience, offers elasticity combined with an optimal level of stiffness. It acts as natural compression, preventing excessive dilation of blood and lymphatic vessels in the limbs. The lightweight design minimizes pressure discomfort while effectively supporting lymphatic and blood flow, which is essential for edema management.
The AM-OSS-16 compression wrap exerts steady, moderate pressure on the tissues, preventing fluid accumulation in the distal areas of the lower limbs. It effectively improves lymphatic and venous circulation, reduces swelling and fluid stagnation, supports post-injury regeneration processes, and decreases the feeling of heaviness and tissue tension.
Its universal design allows the brace to be used on both the left or right foot, while breathable materials ensure comfort throughout the day.
The brace is simple and intuitive to use. It is applied by wrapping it around the foot and ankle requiring compression. The adjustable straps allow the user to modify the level of pressure so that it is effective yet comfortable. Depending on the dynamics of swelling, the compression level may be adjusted throughout the day. It is recommended as part of a compression therapy plan prescribed by a specialist.
Purpose of use:
- Lymphedema of the foot and ankle
- Chronic venous insufficiency
- Varicose veins and venous stasis
- Post-traumatic swelling (sprains, contusions, ligament strain in the foot and ankle)
- Post-surgical, vascular, and orthopaedic conditions
- Edema prevention for individuals who stand or are physically active for long periods
Size table
| Size |
Metatarsal circumference |
How to measure |
| M | 19 – 26 cm | 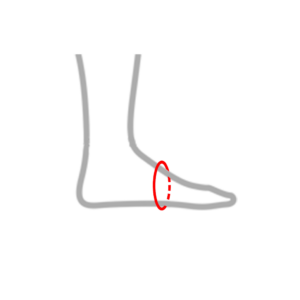 |
| L | 26 – 32 cm |
Right and left foot specific.
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES..
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.




 Class I medical device in accordance with the Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices.
Class I medical device in accordance with the Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017 on medical devices.